  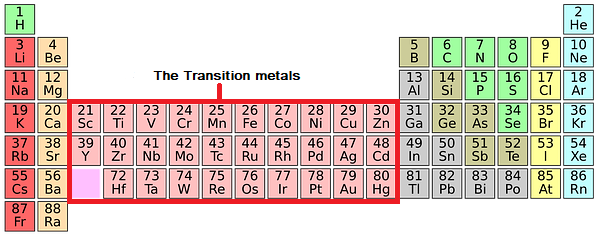 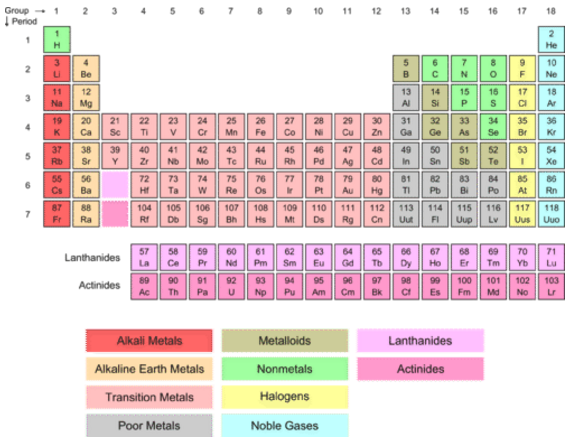
This describes groups 3 through 12 on the periodic table, although the f-block elements (lanthanides and actinides, below the main body of the periodic table) are also transition metals. They can also form Zintl phases (half-metallic compounds formed between highly electropositive metals and moderately electronegative metals or metalloids). According to the IUPAC, a transition metal is any element with a partially filled d electron sub-shell. Being close to the metal-nonmetal border, their crystalline structures tend to show covalent or directional bonding effects, having generally greater complexity or fewer nearest neighbours than other metallic elements.Ĭhemically, they are characterised-to varying degrees-by covalent bonding tendencies, acid-base amphoterism and the formation of anionic species such as aluminates, stannates, and bismuthates (in the case of aluminium, tin, and bismuth, respectively). Physically, post-transition metals are soft (or brittle), have poor mechanical strength, and melting points lower than those of the transition metals most also have boiling points lower than those of transition metals. In Periods 7 and above, all p-block elements are post-transition metals. All of them are p-block elements (which use p orbitals for bonding). All of the transition elements are metals. Any element in groups 3 - 12 in the periodic table. Groups 3 - 12 are in the middle of the periodic table and are the transition elements. These groups contain metals, semimetals, and nonmetals. Post-transition metals in Periods 3 through 6 form a triangular block in Groups 13 through 17. Any element in groups 1, 2, and 13 - 18 in the periodic table. Which elements are counted as post-transition metals depends, in periodic table terms, on where the transition metals are taken to end and where the metalloids or Nonmetals are taken to start. These arise from the changes in the atomic structure of the chemical elements. The strictly periodic table of the elements is. The block names (s, p, d, and f) are derived from the spectroscopic notation for the value of an electrons. Each block is named after its characteristic orbital: s-block, p-block, d-block, and f-block. The term appears to have been first used by Charles Janet. Mainly it includes electronegativity, ionization energy, electron affinity, atomic radii, ionic radius, metallic character, and chemical reactivity. A table of the transition metal elements is constructed to show their dominant electronic features. A block of the periodic table is a set of elements unified by the atomic orbitals their valence electrons or vacancies lie in. Usually included in this category are Gallium, Indium, Thallium, Tin, Lead and Bismuth. Periodic tables are the patterns of the properties of chemical elements that are in the periodic table of elements. In chemistry, are the metallic elements in the periodic table located between the transition metals to their left and the metalloids to their right. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
